Sekisui OSOM COVID-19 Antigen Rapid Test 40-Count EUA
Sekisui OSOM COVID-19 Antigen Rapid Test 40-Count EUA is backordered and will ship as soon as it is back in stock.
Shipping & Returns
Shipping & Returns
Orders are processed promptly and ship from our U.S. warehouse. Shipping costs and delivery times are calculated at checkout based on your location and selected service.
We accept returns on unopened items within 30 days of purchase. Return shipping costs are the responsibility of the customer unless an item is deemed defective.
Some items may be marked final sale and are not eligible for return.

Sekisui OSOM COVID-19 Antigen Rapid Test 40-Count EUA is backordered and will ship as soon as it is back in stock.
Description
Description
- ✔ 40 tests per kit (Cat. 1066-40)
- ✔ EUA authorized for SARS-CoV-2 antigen detection
- ✔ Results in only 15 minutes
- ✔ CLIA-waived for use in professional settings
- ✔ Direct mid-turbinate nasal swab for patient comfort
The OSOM® COVID-19 Antigen Rapid Test is a lateral flow chromatographic immunoassay designed for the qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in direct mid-turbinate nasal swab specimens. It is intended for use by healthcare providers with patients suspected of COVID-19 within the first 7 days of symptom onset.
Authorized under FDA Emergency Use Authorization (EUA), this test offers high positive and negative percent agreement to help reduce the risk of misdiagnosis. Proudly made in the USA, it comes complete with all components needed to run the test without specialized equipment, making it an efficient choice for CLIA-waived settings.
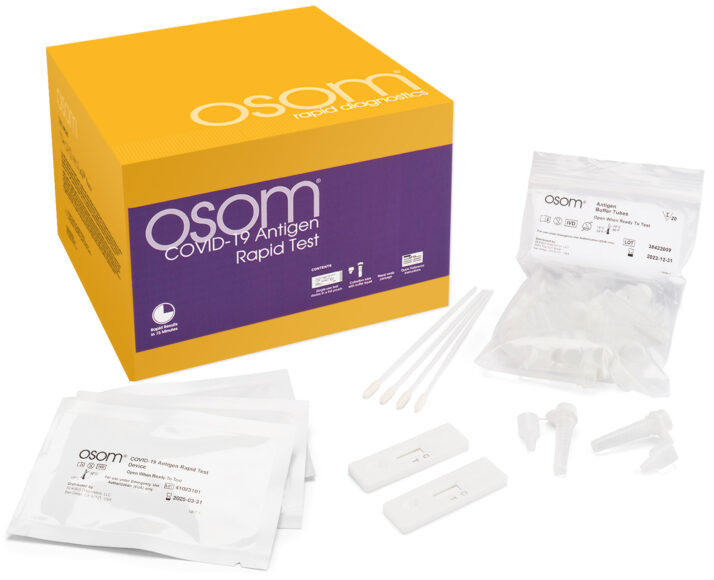
What's in the box
- 40 OSOM® COVID-19 Antigen Rapid Test devices
- Mid-turbinate nasal swabs
- All necessary reagents and accessories
- Package insert & instructions
📄 View Quick Reference Guide (PDF)
For professional use only. Licensing may be required depending on local regulations. Authorized by the FDA under an EUA for the detection of SARS-CoV-2 antigen only, for the duration of the emergency declaration unless terminated or revoked sooner. Item is considered HAZMAT and cannot ship via Air or to AK, GU, HI, PR, or VI. Item is non-returnable.